The University of Nottingham has played host to the launch of a new programme to help East Midlands researchers commercialise life-changing treatments. SPARK The Midlands, the first UK branch of Stanford University's prestigious global SPARK programme – the global pre-clinical accelerator programme with a presence in more than 50 locations...
Upperton Advances Sterile Manufacturing Facility with MHRA Submission
Upperton has formally submitted its application to the UK Medicines and Healthcare products Regulatory Agency (MHRA) for approval of its 7,000 sq. ft sterile manufacturing facility. This submission follows the successful completion of aseptic process simulation (APS) validation batches executed under the stringent requirements of the revised EU GMP Annex...
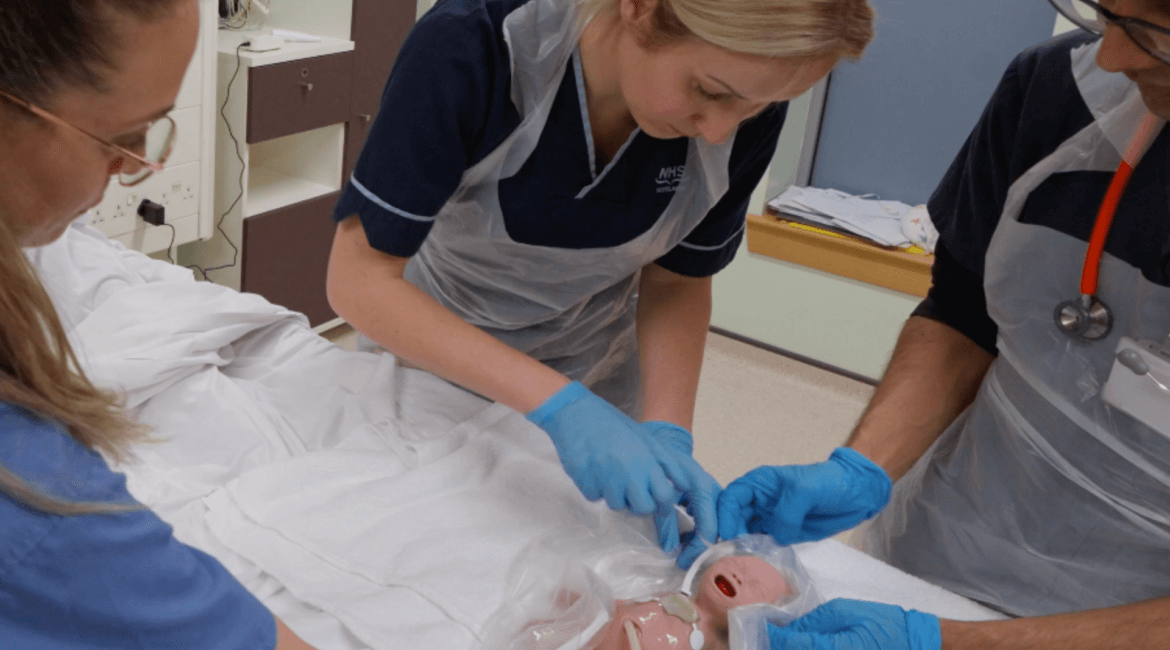
Advancing Neonatal Monitoring with MeTAP Support – Medilink Midlands Case Study
At a glance: Company: SurePulse Sector: MedTech / Neonatal Care MeTAP support accessed: Grant funding, clinical simulation coordination, neonatal expertise, video capture and advisory support Key outcome: Workflow validation of SurePulse NeoPatch, quantifiable efficiency gains, regulatory progression support The challenge: SurePulse is developing SurePulse NeoPatch, a wireless, multi-parameter neonatal monitoring...
Medilink Midlands announces finalists for the 2026 Business Awards
Medilink Midlands is delighted to announce the finalists for the 2026 Medilink Midlands Business Awards, celebrating excellence, innovation, and leadership across the region’s life sciences and medtech sectors. The awards recognise companies, universities, and healthcare innovators driving growth, improving patient outcomes, and delivering cutting-edge solutions in health and care. The...
Shaping West Midlands health tech together. Join the next stage of the North Star journey
The West Midlands Health Technologies Cluster (WMHTC) is defining its next chapter - and there’s still time for leaders across industry, academia, and the public sector to get involved. Workshop 1: Insight & Evidence started the conversation, reviewing regional insights, exploring best-practice examples, and identifying key opportunities and challenges for...
Black Space Technology demonstrates telemedicine capability on Everest
Midlands-based Black Space Technology supported a landmark Mount Everest expedition that saw the first mixed gender team of British Army Reservists successfully reach the summit of Everest. Through the deployment of its Rapid EPR telemedicine kit, the company demonstrated how advanced medical technology can enable first-class delivery of healthcare into...
HORIBA to Build a New Global Headquarters
Advancing global operations and realigning domestic sites to strengthen the organisation and lay the groundwork to become a company with annual revenue of 1 trillion yen. HORIBA, Ltd. (Head Office: Kyoto, Japan; President: Masayuki Adachi) announces plans to build a new global headquarters on the site of its current head...
MHRA launches a consultation on indefinite recognition of CE-marked medical devices
The Medicines and Healthcare products Regulatory Agency (MHRA) has today launched a consultation on proposals for indefinite recognition of CE-marked medical devices in Great Britain (GB), aimed at protecting patient access and ensuring the continued supply of safe and effective medical technologies. Around 90% of medical devices currently used in Great Britain are CE...
AI takes healthcare by storm: understanding how to safely launch devices in the UK, US and EU
A new whitepaper by regulatory experts IMed Group and life science software developers Firefinch Software Oxford, 12th February 2026 – IMed Group, a leading provider of regulatory consultancy services to the global medical and health technology industry, and Firefinch Software, software a leading life sciences software development agency based in...
The Scientific Laboratory Show & Conference 2026 Returns to Nottingham
The Scientific Laboratory Show & Conference 2026 is a trade show like no other, brought to you by Scientific Laboratory Supplies (SLS), the UK’s largest independent supplier of laboratory equipment, chemicals and consumables returns to Nottingham on Wednesday 13th May 2026 following the record-breaking success of the 2024 show, which...