Paul Edmondson and Rouzet Agaiby will join the eg technology board of directors, who are responsible for the strategic direction of the company as well as overseeing, planning and managing the implementation of daily operations. The board is also accountable to the employee ownership trust. Paul has been with eg...
eg technology launch new eBook – Connected Devices: A guide to developing the right product
The value of a well-managed connected device can be significant, to both individuals and businesses. So, it is no surprise that the connected device ecosystem is not only thriving but growing exponentially. In fact, McKinsey Global Institute is estimating that connected (IoT) products and services will generate between $5.5 trillion...
Pioneer Group and Innovate UK join forces on programme aiming to accelerate the development of life sciences companies
Pioneer Group, a company combining laboratory development and operation with venture building, is continuing its collaboration with Innovate UK by bringing its venture development ‘Launch Programme’ to the Exploit stage of Innovate UK’s ICURe programme. The Launch Programme welcomes invited life science founders from the ICURe Programme, as well as...
Penningtons Manches Cooper appointed as legal advisor to Advanced Research + Invention Agency (ARIA)
Penningtons Manches Cooper’s Birmingham and Oxford offices have been appointed as legal advisors to the Advanced Research + Invention Agency (ARIA), a new R&D agency built to unlock scientific and technological breakthroughs to benefit everyone. ARIA was formed in January 2023. Backed by an £800 million investment from the UK...
Cambridgeshire engineering firm appoints new director
A Chatteris-based engineering company has announced the appointment of a new director. Advanced Precision Technologies, which specialises in 5-axis CNC programming and manufacturing, has appointed Danny Speirs as a co-director, signalling the start of a phase of planned expansion. Danny brings with him 25 years of manufacturing engineering experience and...
Leveraging Your CE Mark With a Certificate of Free Sale
Are you leveraging your CE mark in order to make your medical devices & IVD devices available in markets outside the European Union (EU)? In many of these markets a Certificate of Free Sale is required to demonstrate EU compliance and forms part of the application process. The medical device...
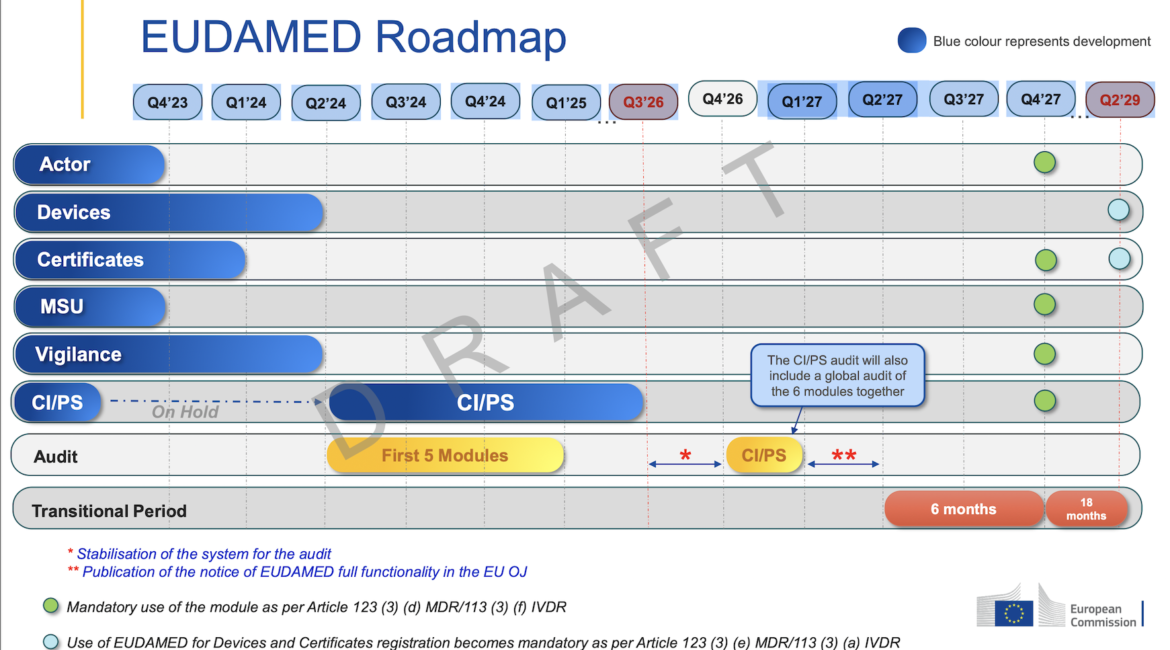
Mandatory Implementation of EUDAMED Delayed Once Again
The EU Commission seemed to have confirmed a delay to the mandatory implementation of EUDAMED by posting a new draft timeline to its website. The draft timeline indicates that all six of the modules will not be in a position to undergo independent audits before Q4 of 2026, this is...
Risk under the microscope: Five challenges facing the life sciences sector
2023 is a pivotal year for UK life sciences as the Government renewed its commitment to increasing funding for research and development, and signed a new deal to re-join the European Union’s €95.5 billion research funding programme known as Horizon Europe. In January, George Freeman, Minister for Science, Research and...
West Midlands duo secures £1.5m Indian export order
Two West Midlands manufacturers are marking 40 years of collaboration by securing an export order at Blechexpo. Brandauer, a leading independent metal stamper, and Auric Metal Finishers from Coventry have partnered to design, develop, and supply customised EloPin push fit-connectors for a tier 1 automotive supplier in India. This £1.5m...
Medilink Midlands celebrates 20 years of supporting businesses
Innovation is the key for growth within the healthcare and Life Sciences sector. Without innovation the sector would stagnate, so it's important that companies and innovators are supported with their great ideas and help them get on their pathway from concept to commercialisation. At Medilink Midlands we are celebrating our...